You can learn more about MDLs here and find out why most medical device cases are not suited for class action lawsuits. Bard have been consolidated into two separate multidistrict litigations (MDLs) to help the lawsuits resolve quickly. That being said, the lawsuits against Cook Medical and C.R. You are not automatically covered by the pending litigation. Tragically, the person had to have a second open-heart surgery procedure in order for the. If you or a loved one suffered injuries from an IVC filter, you will need to file your own, individual lawsuit with an attorney of your choice. It was a person who had been given an implant prior to having surgery. 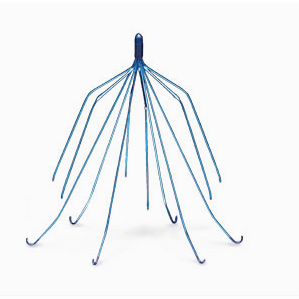
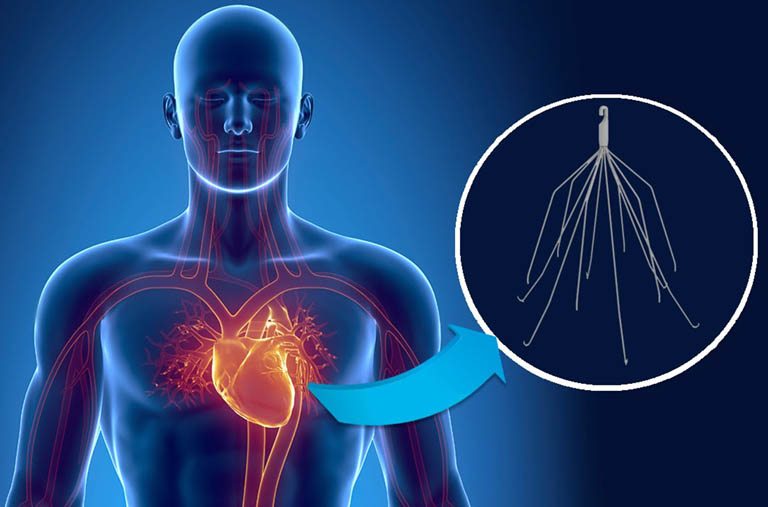
The suits allege that both Bard and Cook Medical knew that its IVC filters weren’t fit for use, but failed to recall them or warn patients about the serious risks associated with the devices. Puncturing of the aorta, duodenum, small bowel, ureter or spine.Cardiac tamponade (pressure caused by fluid build-up in the sac surrounding the heart).Our legal team has decades of experience and will work quickly to obtain the maximum amount of compensation for your IVC filter lawsuit.Ĭall 1 (855) 722-2552 or fill out a contact form to get started. Bard IVC filter lawsuit or a Cook Medical IVC filter lawsuit, you can hold these companies. If you or a loved on has suffered from any of the above IVC filter complications or side effects, you may be able to recover compensation for medical bills, lost wages, pain and suffering and other damages related to the dangerous medical device through an IVC filter lawsuit.įor immediate help from the team of experienced class action lawyers at Gordon & Partners, contact us for a FREE, no obligation consultation. In addition to gaining compensation for damages, by filing a C.R. Some IVC filter types include the Bard Recovery and G2 IVC filters, which are manufactured by C.R. A Pulmonary Embolism (PE) is a severe and often fatal condition triggered when a lower extremity thrombus (blood clot) moves. IVC Filter Injuries, Pulmonary Embolism, Side Effects. Lawsuits targeting Bard and its subsidiary companies have been filed and include allegations of design and manufacturing defects, negligent misrepresentation, breach of implied warranty, negligence and failure to warn. We will keep you up to date on any IVC filter class action lawsuits, additional FDA IVC Filter warnings, and IVC Filter FDA recall announcements. These metal pieces can wreak havoc and cause serious injuries or even death. A Pulmonary Embolism (PE) is a severe and often fatal condition triggered when a lower extremity thrombus (blood clot) moves to the lungs. However, some filters often remain in place until they fall apart in the form of metal pieces or splinters. We will keep you up to date on any IVC filter class action lawsuits, additional FDA IVC Filter warnings, and IVC Filter FDA recall announcements. Since 2005, the FDA has received 921 device adverse event reports involving IVC filters. Most of these filters are retrievable and are not designed to be left in the body indefinitely. 
Placed in the Inferior Vena Cava, Bard IVC filters prevent blood clots that may travel to the heart, lungs or brain from forming. If your medical complications were caused by a Bard IVC filter, you may be entitled to file a claim for damages. 
The company also manufactured the recovery cone removal system, used to retrieve IVC filters, without the required clearance or approval and failed to inform the agency of device malfunctions. Bard's locations in New York and Arizona misfiled consumer complaints, including the report of a patient death. In 2015, the FDA issued a warning letter that C.R. Patients, who no longer need these filters in place because their risk of pulmonary embolism is gone should have their filters removed, according to the FDA.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
